A variety of contaminants, which are either chemical elements or compounds, enter a gas turbine engine. Some of the contaminants to gas turbines that are detrimental are Sodium, Potassium, Vanadium Sulfur, Lead, Chlorine, Fluorine, Magnesium, Calcium, Silicon, Nitrogen, Water, Sediments and Inert Particulates. Unusual contaminants such as Mercury, Cadmium, Bismuth, Arsenic, Indium, Antimony, Phosphorous, Boron, Gallium, etc., present above a certain threshold concentration would require special treatment and precautions, as per the turbine supplier’s recommendations.
Contaminants: Air, Water and Fuel
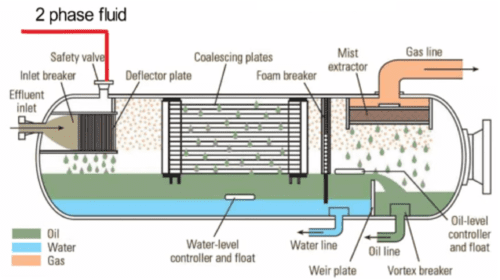
Air: Gas turbines operate in all types of geographical and physical locations worldwide. Airborne contaminants often are site specific and include sea salt, dust and sand, factory discharge gases, exhaust fumes containing oil and fuel vapors, particulates such as chemicals, fertilizers, mineral ores and any variety of industrial by-products. Airborne contaminants can vary daily and/or seasonally. They are subject to climatic conditions such as prevailing wind direction, wind speed, temperature, relative humidity and precipitation. Inlet air contaminants find their way into the compressor inlet and move through the turbine gas path. The most important consideration in selecting an air filter is the particulate size and composition in the incoming air. The incoming air can contain contaminants from many sources, depending on the site’s location. In this article, the focus is on a coastal site which is likely to have significant amounts of alkali salts (sodium and potassium). The contaminants carried by air in marine, offshore and coastal environments are:
A seawater sample of 35 oz contains salt – 1.23 oz which is comprised of the constituents:
- Chloride 55%
- Sodium 30.6%
- Sulfate 7.7%
- Magnesium 3.7%
- Calcium 1.2%
- Potassium 1.1%
- Other constituents 0.7%
Any airborne particulate larger than 5 microns causes erosion to compressor blades and vanes.
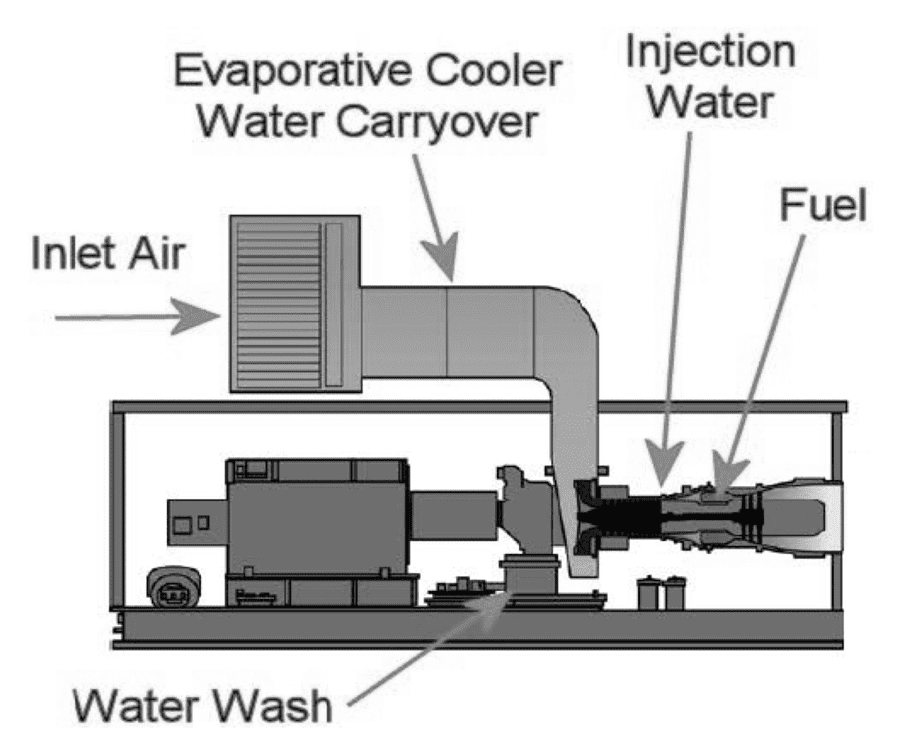
Water: Water is used in turbines for three primary purposes: 1) evaporative cooling to increase power when operating in high ambient temperature conditions, 2) water washing of the compressor section and 3) suppression of NOx emissions. Evaporative coolers are employed with gas turbine engines to reduce the inlet air temperature and thereby increase power output. They are most effective in hot, dry environments. As inlet air flows through the evaporative cooler media, the resulting water evaporation extracts heat from the air, thus cooling it. With these systems, there is potential for water carryover into the turbine. Although proper design (e.g., vane type mist eliminators), installation, operation and maintenance can minimize the amount of water carryover, it is recognized that some amount of water will carry over from the evaporative cooler in the form of water droplets of varying size. The water carryover contains some water-soluble contaminants originally found in the inlet air and dissolved in the water during passage of the air through the evaporative cooler. Compressor cleaning usually involves water-based solutions being introduced into the engine. The compressor deposits removed by these solutions travel through the turbine and exit either through the combustor housing drain or through the exhaust. Water can be injected into the combustor for NOx emission control purposes. This water will travel through the turbine hot section gas path. Although water consumption is typically somewhat less than fuel consumption, the total volume of water introduced into the engine can be quite large. Last, but not least, water is used in small amounts from time to time to purge liquid fuel passages in dual fuel injectors during fuel transfers and liquid fuel shutdown. Water used for all these purposes, if not properly treated, can often introduce sodium, potassium or other contaminants into the turbine leading to higher risks of corrosion.
Fuel: Gas turbine engines can normally operate on dual fuel (gaseous or liquid fuels). Gaseous fuels include natural gas, digester gas, landfill gas, associated gas, coal gas or syngas and coke oven gas. Natural gas is the most common fuel used for industrial gas turbines. It consists primarily of methane with small amounts of inert substances, such as nitrogen and carbon dioxide, and heavier hydrocarbons such as ethane and propane. These heavier hydrocarbons are sometimes used as fuels by themselves or in mixtures. The contaminant of greatest concern that may be found in natural gas is hydrogen sulfide. If the gas is contaminated with water, it may also contain alkali metals. The four main types of liquid fuels are crudes, distillates, residuals and non-hydrocarbons. Distillates and residual fuels are obtained by the refining of crudes. Therefore, the contaminants found in distillates and residuals originate from the crude feedstock or mishandling during processing or transportation. The liquid fuel most frequently used in gas turbine applications is high speed diesel. Different specifications for this fuel limit the amount of sulfur to certain levels. These limits differ in various countries and parts of the world. In addition, diesel fuel may be subject to further contamination during transportation.
Contaminant Effects
Contaminants can affect gas turbine engine components in three ways: fouling/deposition, erosion and corrosion. Fouling/deposition and erosion are physical phenomena induced by the ingestion of chemically inert solid particulates. Corrosion includes a variety of chemical phenomena induced by the reaction of contaminants with turbine engine components. Of the contaminant effects, corrosion has the greatest impact on power output and durability for industrial gas turbines.
Gas Turbine Corrosion Mechanisms
Ingested contaminants can result in corrosion to the compressor, combustion and turbine sections of gas turbine engines. In most applications, corrosion of compressor components is unlikely during engine operation because the compressor is dry. However, during shutdowns where cold surfaces condense water, chemical species such as hydrochloric acid and sulfur trioxide can be absorbed in the water producing an acidic, corrosive liquid. This liquid phase can result in aqueous corrosion of compressor components through a variety of mechanisms, e.g., generalized, pitting and crevice corrosion, and stress corrosion cracking. In cases where hydrochloric acid and sulfur trioxide are present, and the relative humidity is high, the high velocity of the air at the compressor inlet causes the temperature of the air to drop due to conversion of internal energy to kinetic energy. The temperature drop can result in the formation of a liquid phase in the forward stages of the compressor during operation. Aqueous/ acidic corrosion of fuel delivery system components are the two main types of corrosion that are known to affect combustion section components, such as fuel manifolds, fuel flow dividers and fuel injector braze joints. This corrosion can occur in much the same way as the aqueous corrosion of compressor components, except that the contaminants can also be fuel borne. Corrosion to these components can result in fuel leaks and fires, and malfunction of fuel injectors. Sulfidation, which is the reaction between a metal along with sulfur and oxygen-containing atmosphere to form sulfides and/or oxides, can affect fuel injector tips. In essence, sulfidation attack is a form of accelerated oxidation resulting in rapid degradation of the substrate material due to loss of corrosion protection. Whereas during oxidation, protective oxide scales can be formed, the metallic sulfides formed are not protective. This accounts for the rapid rate of degradation produced by sulfidation attacks. Hot corrosion is the most serious form of corrosion experienced by the turbine section components. To better understand hot corrosion, it is useful to first discuss oxidation. Oxidation is the chemical reaction at high temperatures between a component and the oxygen in its surrounding gaseous environment. Oxidation of turbine section components is relatively easy to predict, and measures can be taken to control it, since it primarily involves relatively simple metal/oxygen reactions. The oxidation rate increases with temperature. Metal loss due to oxidation can be reduced by the formation of protective oxide scales. Chromium, aluminum and silicon are the only chemical elements known to form protective oxide scales at the temperatures encountered in gas turbine engine hot sections. The presence of these elements in turbine engine alloys and coatings results in improved oxidation resistance. Hot corrosion is a form of accelerated oxidation that is produced by the chemical reaction between a component and molten salts deposited on its surface. Hot corrosion comprises a complex series of chemical reactions, making corrosion rates very difficult to predict. Sodium sulfate is usually the primary component of the deposit; degradation becomes more severe with increasing concentration levels of contaminants such as sodium, potassium, vanadium, sulfur, chlorine, fluorine and lead. The rate and mechanism of hot corrosion attack is influenced by temperature. There are two types of hot corrosion. Type I, or high temperature, hot corrosion occurs at a temperature range of 1346 to 1742°F. Type II, or low temperature, hot corrosion occurs at a temperature range of 1022 to 1346°F. Both types can occur in the turbine section.


Prevention and Mitigation
There are three categories of environments to consider in relation to hot corrosion: marine, offshore and coastal. Salt is present in all of these environments, and it is critical to stop it from entering the turbine where it can cause corrosion. A site environmental analysis survey should be performed at the earliest opportunity in the project planning process to understand the conditions that affect filtration selection. There are inland sites where significant amounts of salt are present, such as certain deserts, dried lakes and certain industrial sites. These should be treated as coastal when salt levels are determined to be significant. [Coastal – This classification includes any land-based application within 10 miles of a coastline. Weather conditions can run from dry and sunny to rain, snow, sea mist and freezing fog. Blowing sand, industrial dusts and unburned hydrocarbons are present in many coastal environments. Tropical conditions such as heavy rains, high humidity and large quantities of insects can also occur. High velocity filters are not recommended for any coastal application due to their low dust holding capability. Medium velocity (for better protection against smaller particles in areas with humidity below 50 percent) or self-cleaning filters should be used, depending on the environment.]
Gas Turbine Material Selection: Control of contaminants to concentrations below the desired limits (which is project specific and environment specific), the preferred path toward trouble-free operation, and other material solutions are taken to mitigate the effects of gas turbine engine hot corrosion. The selection of materials, e.g., alloys and coatings, should take into account typical operating conditions and should allow for some occasional system upsets. Corrosion resistance is one of the attributes considered while selecting materials. In situations where the corrosion resistance of alloys is deemed insufficient for the application, protective coatings are specified. Superalloys, such as 718 and 901, used in the compressor section do not require protective coatings. Stainless steels such as 410 and 17-4PH are commonly used for compressor stationary and rotating applications. Although these alloys have inherently good corrosion resistance, a protective coating is commonly used on compressor components made from these alloys to enhance their durability. Turbine section airfoils, e.g., blades and nozzles, are the components most susceptible to hot corrosion attack. Turbine section airfoil components are thus considered systems in which the base alloy provides the mechanical and structural capabilities, and the coatings contribute the resistance to oxidation and hot corrosion attack. Coatings, such as silicon aluminides, for under-the-platform regions of the turbine blades may be required for severe conditions (Type II hot corrosion regime). High chromium weld materials, such as IN-738, may be applied to turbine blade tips to enhance blade tip hot corrosion resistance (Type I hot corrosion regime).
Conclusion
The proper air filtration, fuel handling and treatment selections can be specified to keep contaminants at or below acceptable levels. With careful management of the contaminants that can cause corrosion, gas turbines have proven to be reliable and robust prime movers that can be counted on to provide years of cost-effective service.
Nirmal Surendran Menon, PMP®, is the author of the book, Valves Made Easy, along with a coauthor, who is an industry expert as well as a project management professional with 17-plus years of experience in the oil and gas/energy facility construction. He currently works as a senior construction manager overseeing the Pipe Pressure Testing and Pipe Cleaning portfolio with an EPC company with a distinguished reputation in the industry. Menon’s interests include pipe integrity, pipe cleanliness, loss prevention, and human factors for process facilities. He has authored 11 articles and two books, and has sustained national and international acclaim in the industry, having been part of major LNG project construction execution in different geographical locations.
Oil and gas operations are commonly found in remote locations far from company headquarters. Now, it's possible to monitor pump operations, collate and analyze seismic data, and track employees around the world from almost anywhere. Whether employees are in the office or in the field, the internet and related applications enable a greater multidirectional flow of information – and control – than ever before.